Why Supplier Data Isn't Enough to Substantiate Your Efficacy Claims
Most brands substantiating efficacy claims lean on data provided by their ingredient suppliers. On the surface, this seems reasonable…the supplier conducted the research, the ingredient is in the formula, so the claim should hold. For softer, more “fluff” claims this approach may be passable, but for stronger efficacy claims supplier data alone could get you in hot water.
The truth that most brands don’t want to hear is that data generated from testing one formula cannot just be universally applied to any formula.
Why?
Well, that is because the efficacy of the active is directly shaped by the properties of the formulation it's sitting in.
If your formulation doesn’t behave exactly like the supplier’s, you may not see the same results. That means that the data you have used to back up that anti-wrinkle claim won’t hold up because you have hindered that ingredients ability to provide any anti-wrinkle benefit.
A supplier study tells you what's possible with the ingredient in that vehicle, but it doesn’t really say anything about what your formula will actually deliver.
Properties that Control Active Absorption
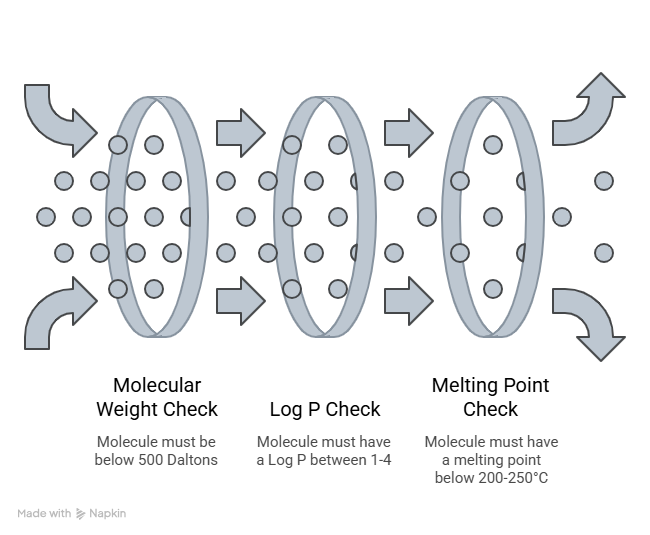
Before any active can do its job, it has to get through the stratum corneum (SC). Since the SC is the main hurdle any molecule has to overcome, this is a crucial step that can determine an actives fate. The intact SC is generally permeable only to compounds with a…
Molecular weight below 500 Daltons
Log P between 1-4
Melting point below 200-250C
The key step that will determine an active’s fate is the penetration into the SC. This requires the solvent that the active is dissolved in to partition out of the formulation and into the stratum corneum. Next, the active has to leave the solvent and move into the SC.
This process is a delicate balance between the molecular properties of the drug, the skin properties of the user, and the formulation properties. Together, these drive the interactions between the active-vehicle, vehicle-skin, and active-skin which all contribute to the active partitioning into the SC.
The choice of formulation ingredients can influence this key step through several mechanisms simultaneously. The base will control how much of the solvent (that the active is dissolved in) is available to partition into the SC, whether it alters the SC's barrier properties, and how well the active is released from the solvent once in the skin. Change one ingredient and you may be shifting several of these variables at once, without realizing it.
When a supplier runs an efficacy study on their active, they test it in a vehicle of their choosing, which is usually a simple base designed to showcase the ingredient's performance under ideal conditions. That base is almost certainly different from yours with a different emulsifier, different oil phase, different rheology modifiers, different pH, etc.
The differences between the supplier's base and yours could hinder penetration and permeation of the active, which could ultimately mean that your formula isn't delivering what you think it is.
Still don’t believe me? Let’s take a look at a few case studies.
How the Base Formula Can Affect Active Penetration and Permeation
Case Study 1: Penetration Enhancers
Caffeine is one of the most common actives in eye creams, routinely appearing on ingredient lists alongside claims about puffiness, dark circles, and fine lines.
A 2023 study published by Reyes et al. set out to design and optimize caffeine eye creams using Hansen Solubility Parameters (HSPs), then benchmark them against three commercially available caffeine eye creams [1].
They found that the commercial products released only 11–19% of their caffeine over 24 hours while their formulations released over 85%. In their permeation study, they also found that two of their formulations EC3 and EC4 significantly outperformed the rest with 34% cumulative percent of caffeine permeated per unit area [1].
Why such a gap? Well, the authors note that the amount and type of solvent, the solubility of the caffeine in the formulation, the pH, the base ingredients and the interactions among the ingredients can all affect the release of a drug.
Caffeine is hydrophilic with a logP of around −0.1, which makes it a difficult candidate for stratum corneum penetration to begin with. A formulation that "traps" caffeine too comfortably in its base means that it likely won’t go far. The commercial products also contained 23 to 57 individual ingredients; as the researchers noted, with more ingredients, the chance of chemical interaction is higher, and additional components may decrease the solubility of caffeine in the product base, limit its penetration, or serve as solvents that reduce its thermodynamic activity, which results in a reduced release and penetration rate [1]. In their formulations the penetration enhancers were carefully selected to work synergistically together to improve caffeine penetration and permeation.
Now let’s imagine you decide to use your supplier’s caffeine data to back up your eye cream’s wrinkle claims…
If a supplier tested their caffeine in a base with favorable solubility parameters and penetration enhancers and your product is a 30+ ingredient emulsion where caffeine could be releasing very poorly, you are not selling the same product. The ingredient might be the same, but the outcome could be drastically different.
Case Study 2: Emollient Selection
When formulators choose emollients, the conversation usually centers on aesthetics such as the skin feel, spreadability, and finish. These are legitimate concerns, but they're incomplete. The emollient system is also one of the variables in your formula that can hinder or improve active penetration, especially for oil soluble actives.
On the viscosity side, research has shown that release from the inner phases of creams is more dependent on the emollient's own viscosity than on the overall bulk viscosity of the finished emulsion [2]. A formulator can look at a finished cream's rheology profile, decide the texture is appropriate, and move on without ever questioning whether the emollient sitting in the oil phase is creating a viscous internal environment that slows the active's escape. As Bolla et al. noted, viscosity can influence the diffusion rate from vehicles and the subsequent drug release. Reformulating for a richer, more indulgent feel, swapping to a heavier emollient, or adding oil soluble thickening agents could change the delivery of the very actives being marketed [2].
Polarity adds a second layer of complexity. Wiechers et al. (2004) laid out the theoretical framework for this clearly: to get an active ingredient to move from a formula into the stratum corneum, the quantity of molecules penetrating into the SC can be increased by increasing the solubility of the penetrant in the stratum corneum, or by reducing its solubility in the formulation [3]. In other words, the active needs to be less comfortable in the vehicle than it is in the SC. That thermodynamic activity is the driving force for getting the active to exit the vehicle and partition into the skin.
The authors introduced the concept of the Relative Polarity Index (RPI): a tool for matching emollient polarity to the active's polarity and the SC's polarity in a deliberate, calculated way. The approach involves a two-step process: first selecting a primary emollient in which the active dissolves well, then introducing a secondary emollient with a sufficiently different polarity to reduce the active's solubility, pushing the formulation toward a polarity far enough from the active to create a driving force toward the skin [3].
The practical consequences of ignoring this were demonstrated head-on. Two formulations containing the same active, dioic acid, a lipophilic skin-brightening ingredient were prepared. One used a standard caprylic/capric triglyceride-based oil phase and the other was designed according to RPI principles using propylene glycol isostearate as the primary emollient and triethylhexanoin to reduce solubility and build the driving force. The delivery-optimized formulation delivered 3.5 times more dioic acid into the skin [3].
The researchers also found that simply increasing the concentration of dioic acid in the cap/cap trig formula from 2% to 8% yielded only a two-fold increase in skin delivery [3]. The conclusion the researchers drew is worth sitting with it… it may be advisable to change a standard formulation by selecting emollients according to the RPI concept rather than change the active ingredient or its concentration.
So, even something as simple as your emollient selection can influence delivery. When you borrow supplier’s efficacy data to substantiate a claim, you're inheriting results from a formula that may have a fundamentally different emollient system than yours.
Case Study 3: Emulsion Structure
The emulsifier doesn't just hold the formula together, it also shapes how much of the active reaches the skin and where in the skin it ends up.
A study by Wiechers et al. showed that your emulsifier system may actually affect where the active ingredient ends up within the skin. They compared emulsions with octadecenedioic acid and different emulsifiers and found that the total skin absorption was similar but the distribution of the active was different between the different emulsifiers[4].
An active that needs to reach the viable epidermis to trigger a keratinocyte response has a completely different target depth than one that needs to accumulate in the dermis to stimulate fibroblasts. If your emulsifier system is hindering the distribution of your active, then the biological response the supplier demonstrated may never be triggered.
Research by Oborska et al. incorporating polyoxyethylene cetostearyl ethers of varying oxyethylene chain lengths into otherwise identical O/W emulsions and found that with increasing oxyethylene chain length, the permeability of the drug decreased. In this study, the emulsifier's molecular structure was affecting delivery without any change to the active, its concentration, or the rest of the formula [4].
So, if subtle changes to the emulsifier within an O/W emulsion can have different outcomes, you can’t know for certain that your formula will produce the same effect that the supplier’s emulsion did.
Final Thoughts
The evidence across these case studies points to one unavoidable conclusion: an active ingredient is only as effective as the formula it's sitting in.
The caffeine data showed us that formulas containing the same ingredient can deliver anywhere from 11% to over 85% of that ingredient over 24 hours, a difference driven entirely by formulation decisions.
The emollient work demonstrated that polarity mismatches and viscosity characteristics can alter penetration efficiency.
The emulsifier research showed that even when total delivery is equivalent, the distribution of an active across skin compartments can be different all because of a single ingredient swap.
“Use supplier data as a starting point, not an endpoint.”
Supplier studies are useful for establishing what an active is theoretically capable of, such as its mechanism of action, the concentrations at which it has been shown to be effective, and the biological endpoints it can influence. The mistake is treating that data as if it transfers automatically to your formula. Instead, use it to frame your hypothesis: this ingredient, at this concentration, in a formula designed to support its delivery, should be capable of producing this effect.
Then build the evidence for the second half of that sentence yourself.
References
[1] Skin penetration of caffeine from commercial eye creams and eye creams designed and optimized based on Hansen solubility parameters - ScienceDirect
[2] Emollients in dermatological creams: early evaluation for tailoring formulation and therapeutic performance
[3] Formulating for efficacy1 - Wiechers - 2004 - International Journal of Cosmetic Science - Wiley Online Library
[4] Formulation effects of topical emulsions on transdermal and dermal delivery - Otto - 2009 - International Journal of Cosmetic Science - Wiley Online Library