A Primer on Micelle Formation and Packing Parameters of Surfactants
In this month’s Formulation Reading Club deep dive, I am going to uncover what I learned from the surfactant reading list from February.
If you are new here, welcome to the Formulation Reading Club. A monthly reading challenge designed to get you reading, learning something new and joining in on the discussion.
Each month, I share a small, intentional reading list focused on a formulation science topic to help you build on your foundational knowledge, think critically about the research that shapes our industry and stay up to date.
Want to join the reading club?
Sign up below to get the reading list sent to your inbox every month!
The Basics of Micelle Formation
Understanding the microstructure of the surfactants in our formula can help us better control things like viscosity, foaming, oil loading, drug delivery, skin penetration and more. So, let’s dive into how they form and what governs them.
When we add a surfactant to a beaker of water, the molecules will begin to align at the air-water interface. The hydrophilic head group will be positioned towards the water and the hydrophobic tail will be aligned towards the air. At some point, no more monomers will be able to fit at this interface which forces some of them into the bulk solution. The hydrophobic regions are really uncomfortable with this because they don’t like water. So, they spontaneously arrange themselves in a way that reduces their exposure to it. This structure is called a micelle and the concentration at which the micelles form is called the critical micelle concentration (CMC).
Micelles are composed of around 50-100 molecules and typical spherical micelles have the hydrophobic tails oriented inward to create a water free region inside. Micelles can come in different shapes and sizes that are inherently influenced by the molecular structure of the surfactant.
The Packing Parameter: Predicting Microstructure
The structure of a surfactant molecule dictates how it packs with neighboring molecules, and that packing influences things like viscosity, foam, etc.
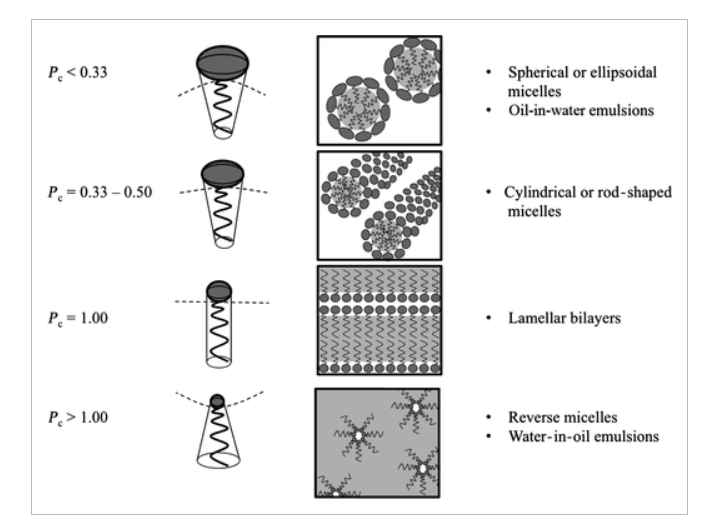
We can define a packing parameter to predict how any given surfactant will behave [2]:
P= v/ Lc ah
Where:
P= packing parameter
V= volume of hydrocarbon tail
Lc= length of tail
ah= cross sectional area of head group
From the above equation, you can start to see how the molecular structure will influence the packing.
If the volume of the tail is increased → the packing parameter will increase
If you decrease the head group area → the packing parameter will increase
If you increase the length of the tail → the packing parameter will decrease
These shifts in P correspond to predictable changes in micelle shape that you can see illustrated below:
Image Source: A review of shampoo surfactant technology: consumer benefits, raw materials and recent developments - Cornwell - 2018 - International Journal of Cosmetic Science - Wiley Online Library
Why Does this Matter for Formulation?
So why should you care whether your surfactant forms a sphere, a cylinder, or a bilayer? Because microstructure has a direct handle on some of the most important properties in your product.
Spherical micelles, despite being the "simplest" structure, are often exactly what you want. Because they don't entangle, systems dominated by spherical micelles have low viscosity and flow very freely which is ideal for leave-on products like toners and micellar waters.
Cylindrical (wormlike) micelles have significantly more opportunity to entangle with one another compared to spherical ones. This entanglement increases the bulk viscosity of the solution, which is a property that matters enormously in products like shampoos and body washes. A thicker feel is not only more pleasant for consumers, but studies have also shown it can improve foam stability.
Lamellar bilayers bring a different set of benefits. In emulsions, they've been shown to be milder on the skin and to improve drug delivery. In rinse-off systems like body washes, they can stabilize high oil loads without significantly compromising foam quality, and they easily suspend particulates like beads and powders.
Understanding the structure you're targeting should inform your surfactant selection from the very beginning. For example, if you want to form lamellar bilayers, you may need to pick a specific blend of surfactants like, Iselux SLC, to achieve this microstructure. These commercially available blends have already done the hard work of choosing the right surfactants at the right concentrations in order to provide the critical packing parameter needed for forming lamellar phases.
But let’s dive into some additional tools to help us manipulate the microstructure.
Surfactant Concentration
First and foremost, microstructure change can be driven by the concentration of the surfactant. For many anionic surfactants, as the concentration increases, the phase behavior changes from spherical to rods to hexagonal to lamellar to inverse hexagonal to inverse micelles [1]. But for practical purposes, increasing the surfactant concentration to manipulate the phase behavior is often not possible due to the extremely high surfactant levels that would be needed (hello skin irritation). So, we need to rely on other methods that allow us to manipulate the packing parameter in more dilute solutions.
Electrolytes (Salt)
One of the most common methods for altering the packing parameter is the use of salt. Since salt will dissociate in solution into the sodium and chloride ions, those sodium ions can interact with the negatively charged head group of anionic surfactants. This creates a shield that allows the head groups to get closer together, reducing the head group area. This reduction in area is going to increase the packing parameter. From our chart above, this means that the micelle shape can change from the spherical micelle to the more wormlike micelle which will result in an increased viscosity. Now, this effect is only possible with strong anionic surfactants (ex. sodium lauryl sulfate) and is concentration dependent. The viscosity will increase as the concentration of salt increases to a certain max concentration. Above this concentration you will start to see a trend back down in viscosity.
If you are working with a surfactant system that you are thickening with salt, make sure you create a salt curve. Test the viscosity of your formula at varying salt concentrations to understand when it peaks and starts to drop back down. You’ll want to stay on the ascending side so that it can be easier to adjust. Remember that most surfactants also have some level of sodium chloride as an impurity.
pH
pH can have an effect in surfactants that have ionizable groups. If the ionization state of a surfactant can vary depending on the solution’s pH, then you may be able to manipulate the packing by altering the charge. Reducing the charge, will reduce the size and ultimately the way the molecules can pack together. This can lead to those shifts in the microstructure.
Cosurfactants, Cosolvents, and Other Additives
Blending two or more surfactants is a highly effective way to tune packing parameters across a wider range than any single surfactant can achieve. A common strategy is pairing a primary anionic surfactant with a zwitterionic cosurfactant, which nudges the blend toward cylindrical micelles because of mixed micelle formation. This is why you see cocamidopropyl betaine and sodium lauryl sulfate used together quite frequently in body washes and shampoos. Other studies have shown that adding low concentrations of a cationic surfactant to an anionic surfactant solution can shift the system towards cylindrical micelles based on the same mechanism as salt [3]. The addition of polymers can also have similar effects. Other studies have shown that the additional of certain co-solvents can affect the micelle structure potentially because of changes to the dielectric constant of the solution, where a decrease in the dielectric constant may increase the interaction between the head groups [3].
Final Thoughts
Most consumers will never think about what's happening at the microscale when they lather up a shampoo. They just know it feels right or it doesn't. But what they're actually responding to is the microstructure you engineered. By understanding the packing parameter and what drives it, you can make more intentional decisions that will directly shape how the product performs.
This reading list was definitely a good reminder why understanding structural and material properties is so important. Next time you are at the bench trying to figure out how to thicken your body wash, stabilize a greater oil load or reduce the foaming of a leave-on product, don’t forget to think about the micelles!
References
[2] nature_and_properties_of_foaming_surfactants.pdf
[3] Rheology of Cosmetic Products: Surfactant Mesophases, Foams and Emulsions